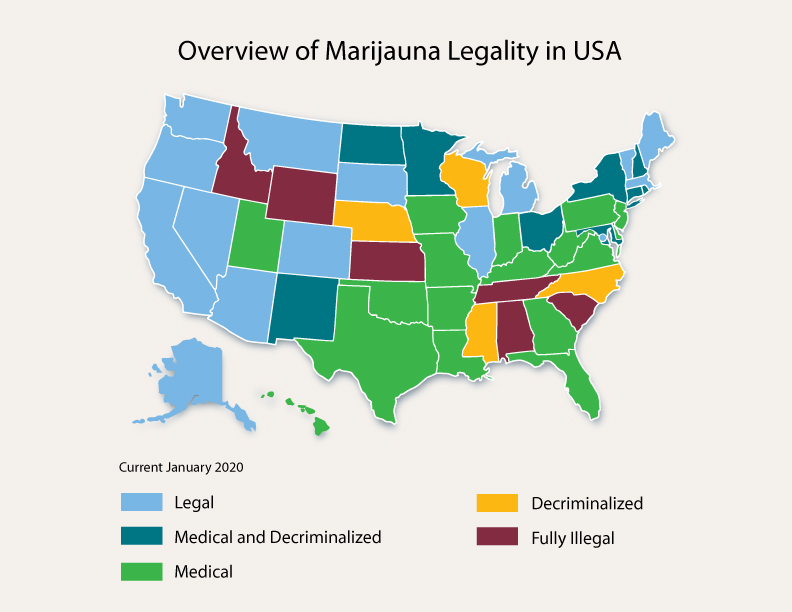
New Hampshire Cannabis Labeling Laws
(a) The dispensing of cannabis or CIP shall be prohibited unless the cannabis is placed within a
container and labeled by an ATC technician in accordance with this section.
(b) All cannabis or CIP to be dispensed shall be packaged as follows:
(1) Cannabis or CIP shall be packaged in plain, opaque, tamper-proof, and child-resistant
containers, except that the container shall not required to be child-resistant if the qualifying
patient or designated caregiver has affirmed in writing that he or she would have difficulty
opening a child-resistant container and that there are no young children living in his or her
household;
(2) Cannabis or CIP shall be packaged in a manner that is not attractive to children;
(3) Depictions of the product, cartoons, or images other than the ATC’s logo shall not be
permitted on the packaging; and
(4) The packaging of edible CIP shall not bear a reasonable resemblance to any
commercially available product.
(c) The ATC shall not use any product name that:
Initial Proposal – Annotated 9/5/14 24
(1) Is identical to, or confusingly similar to, the name of an existing non-cannabis product;
(2) Is identical to, or confusingly similar to, the name of an unlawful product or substance;
(3) Is obscene or indecent;
(4) May encourage the use of cannabis or CIP for recreational purposes;
(6) May encourage the use of cannabis or CIP for a condition other than a debilitating
qualifying medical condition;
(7) Is customarily associated with persons under the age of 18; or
(8) Is related to the benefits, safety or efficacy of the cannabis product unless supported by
substantial evidence or substantial clinical data.
(d) All cannabis or CIP dispensed shall meet the following labeling requirements:
(1) The ATC shall place a legible, firmly affixed label on all cannabis or CIP dispensed,
which contains at a minimum the following information:
a. The qualifying patient’s registry identification number;
b. If the cannabis or CIP is being dispensed to a designated caregiver on behalf of a
qualifying patient, the designated caregiver’s registry identification number shall also
be present;
c. The qualifying patient’s address;
d. The name, registration number, address, and phone number of the ATC that
dispensed the cannabis or CIP;
e. The quantity, in ounces, of usable cannabis dispensed, which, for CIP’s that are
produced by using concentrates, shall be the amount of cannabis used to create the
amount of concentrate included in the CIP;
f. The date and time that the ATC dispensed the cannabis or CIP;
g. The unique batch number of the cannabis or CIP dispensed;
h. The strain(s) of cannabis dispensed or otherwise used in the production of the CIP;
i. The cannabinoid profile of the cannabis or CIP dispensed;
j. This statement, including capitalization: “This product has not been analyzed or
approved by the FDA. It is not certified to be free of contaminants. There is limited
information on the side effects of using this product, and there may be associated health
risks. Do not drive or operate heavy machinery when under the influence of this
product. KEEP THIS PRODUCT AWAY FROM CHILDREN.”; and
Initial Proposal – Annotated 9/5/14 25
k. This statement: “This cannabis or CIP is for therapeutic use only. Diversion of this
product is a class B felony and shall result in the revocation of one’s registry
identification card.”;
(2) In addition to (1) above, the ATC shall place a legible, firmly affixed label on all CIP
including the following information:
a. The type of the product;
b. A list of ingredients;
c. The net weight of the product;
d. A warning if nuts or other “known allergens” are contained in the product;
e. The date of product creation and the recommended “use by” or expiration date;
f. A statement that the CIP, if perishable, must be refrigerated; and
g. Directions for use of the product if relevant;
(3) Labeling text shall not include any false or misleading statements regarding health or
physical benefits to the consumer; and
(4) Multiple labels on a container shall be allowed, however, labels shall be unobstructed
and conspicuous in that no information required by these rules shall be obstructed.
DISCLAIMER: The contents of the Multifeeder Technology website regulatory pages are intended to convey general information only. The information herein should not be relied upon for legal advice on state marijuana laws. We provide links to state cannabis regulatory sites; we do not vouch for or assume any responsibility for the content, accuracy or completeness of material presented in linked sites. The information presented on these pages may not reflect the most current legal developments in the marijuana industry. We disclaim all liability in respect to actions taken or not taken based on any or all of the contents of this site to the fullest extent permitted by law.
NEW HAMPSHIRE STATE MARIJUANA REGULATIONS:
https://www.visit-newhampshire.com/state/marijuana-in-new-hampshire/